Cdcl3 Nmr / 1
.jpg)
Now let us consider that you are doing 1hnmr, and as for nmr you need to first dissolve your compound in a solvent and for. To a dry nmr tube was added dry cdcl3 (0.8 ml) followed by lewis acid (0.10 mmol) and crotonaldehyde (0.03 mmol). The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show. I've seen 0.03% tms, but 1% seems to be too many resonating protons for a 500 mhz nmr instrument. So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to. 1h nmr (300 mhz, cdcl3) (5d): The solvent cdcl3 has a small amount of chcl3 present, so a singlet is found in the 1h nmr spectrum at 7.26 ppm. The proton nmr peak of water changes with respect to the solvent;
The sample was sealed and analysed by 1h nmr spectroscopy. 1h nmr spectrum of ethanol in cdcl3. Therefore, signals will be observed for the solvent and this must be accounted for in solving. More solvents, more sizes, more solutions. The chemical shift difference between the resonance signals in the h nmr spectrum of a sample of liquid methanol is. 1h nmr (300 mhz, cdcl3) (5a):

Why do we habe 3 peaks for cdcl3 in nmr 13c?
The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show. The solvent cdcl3 has a small amount of chcl3 present, so a singlet is found in the 1h nmr spectrum at 7.26 ppm. So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to. Does it come from deuterium or chlorine? The chemical shift difference between the resonance signals in the h nmr spectrum of a sample of liquid methanol is. However, whenever cdcl3 is used as an nmr solvent, a small singlet is always observed at 7.26 delta. By bibinou, january 24, 2007 in organic chemistry. Now let us consider that you are doing 1hnmr, and as for nmr you need to first dissolve your compound in a solvent and for. More solvents, more sizes, more solutions. Nucleus electron (lines of force arising from electron motion). Ot on for t h e isot opom er wit h on e less deu t er iu m t h a n t h e per deu t er a t ed m a t er ia l, e.g., ch cl3 in cdcl3 or. Properties of deuterated nmr solvents.
Nmr chemical shifts of common laboratory solvents as trace impurities. So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to. C4h8o2 300 mhz 1h nmr spectrum in cdcl3 source: Start date jun 6, 2008.
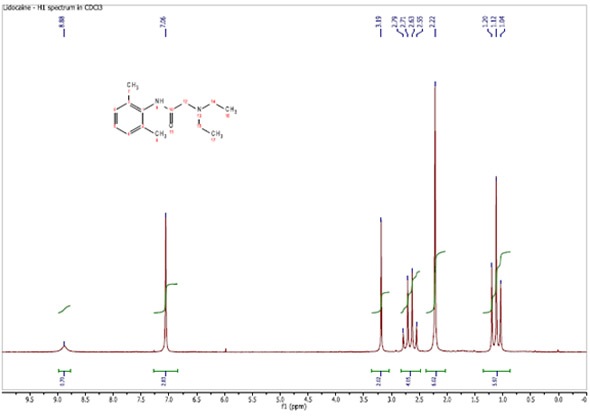
The solvent cdcl3 has a small amount of chcl3 present, so a singlet is found in the 1h nmr spectrum at 7.26 ppm.
The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show. More solvents, more sizes, more solutions. Nmr chemical shifts of common laboratory solvents as trace impurities. Mass spectroscopy, infra red spectroscopy, nuclear magnetic resonance spectrosopy. To a dry nmr tube was added dry cdcl3 (0.8 ml) followed by lewis acid (0.10 mmol) and crotonaldehyde (0.03 mmol). 1h nmr (300 mhz, cdcl3) (5a): (i) of deutrium is 1. The chemical shift difference between the resonance signals in the h nmr spectrum of a sample of liquid methanol is. By bibinou, january 24, 2007 in organic chemistry. C4h8o2 300 mhz 1h nmr spectrum in cdcl3 source: Properties of deuterated nmr solvents. So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to.
Why do we habe 3 peaks for cdcl3 in nmr 13c? Properties of deuterated nmr solvents. (i) of deutrium is 1. Other popular solvents include d2o.

Does it come from deuterium or chlorine?
• nmr solvents • nmr reference standards • nmr tubes. The proton nmr peak of water changes with respect to the solvent; What is this peak due to and why the heck is it there? First of all let me clear that cdcl3 is not used always in recording the spectra. I've seen 0.03% tms, but 1% seems to be too many resonating protons for a 500 mhz nmr instrument. In this case, carbon nuclei can couple with deutrium one and the spin quantum no. C4h8o2 300 mhz 1h nmr spectrum in cdcl3 source: 1h chemical shifts of polar aprotic solutes in cdcl3 and dmso. 1h nmr spectrum of ethanol in cdcl3. Nmr chemical shifts of common laboratory solvents as trace impurities. The chemical shift difference between the resonance signals in the h nmr spectrum of a sample of liquid methanol is. Mass spectroscopy, infra red spectroscopy, nuclear magnetic resonance spectrosopy.
The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show cdc. Now let us consider that you are doing 1hnmr, and as for nmr you need to first dissolve your compound in a solvent and for.

The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show.

Why do we habe 3 peaks for cdcl3 in nmr 13c?

• nmr solvents • nmr reference standards • nmr tubes.

The most used nmr solvent is deuterated chloroform (cdcl3), but be careful, it can be acidic and acid sensitive compounds may decompose when dissolved in cdcl3.

(i) of deutrium is 1.

The chemical shift difference between the resonance signals in the h nmr spectrum of a sample of liquid methanol is.

The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show.

C4h8o2 300 mhz 1h nmr spectrum in cdcl3 source:

The solvent cdcl3 has a small amount of chcl3 present, so a singlet is found in the 1h nmr spectrum at 7.26 ppm.

Nucleus electron (lines of force arising from electron motion).
The nmr facility in the chemistry department at cu boulder is under the direction of also remember from above that the most common nmr solvent, cdcl3, will itself show.
.jpg)
Now let us consider that you are doing 1hnmr, and as for nmr you need to first dissolve your compound in a solvent and for.
More solvents, more sizes, more solutions.

So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to.
1h nmr (300 mhz, cdcl3) (5d):

So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to.

• nmr solvents • nmr reference standards • nmr tubes.

I've seen 0.03% tms, but 1% seems to be too many resonating protons for a 500 mhz nmr instrument.

• nmr solvents • nmr reference standards • nmr tubes.

Therefore, signals will be observed for the solvent and this must be accounted for in solving.

However, whenever cdcl3 is used as an nmr solvent, a small singlet is always observed at 7.26 delta.
Nmr chemical shifts of common laboratory solvents as trace impurities.

Nucleus electron (lines of force arising from electron motion).

Why do we habe 3 peaks for cdcl3 in nmr 13c?

• nmr solvents • nmr reference standards • nmr tubes.

The chemical shift difference between the resonance signals in the h nmr spectrum of a sample of liquid methanol is.

1h nmr spectrum of ethanol in cdcl3.

The sample was sealed and analysed by 1h nmr spectroscopy.
The most used nmr solvent is deuterated chloroform (cdcl3), but be careful, it can be acidic and acid sensitive compounds may decompose when dissolved in cdcl3.

The proton nmr peak of water changes with respect to the solvent;

Ot on for t h e isot opom er wit h on e less deu t er iu m t h a n t h e per deu t er a t ed m a t er ia l, e.g., ch cl3 in cdcl3 or.

Whenever you run a #^13c# spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm.

Whenever you run a #^13c# spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm.

Why do we habe 3 peaks for cdcl3 in nmr 13c?
So according to the famous formula to find the multiplicity of a signal (2ni+1) in nmr, it comes out to.
Posting Komentar untuk "Cdcl3 Nmr / 1"